Powder: -20°C for 3 years | In solvent: -80°C for 1 year
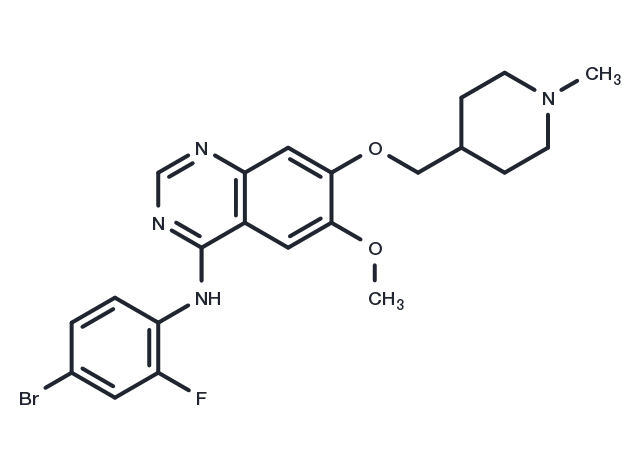
Vandetanib (ZD6474) 是一种具有口服活性的VEGFR2/KDR 酪氨酸激酶抑制剂,IC50值为40 nM。它也抑制VEGFR3/FLT4和EGFR/HER1,IC50值分别为110和500 nM。
| 规格 | 价格/CNY | 货期 | 数量 | |
|---|---|---|---|---|
| 5 mg | ¥ 229 | 现货 | ||
| 10 mg | ¥ 372 | 现货 | ||
| 25 mg | ¥ 494 | 现货 | ||
| 50 mg | ¥ 891 | 现货 | ||
| 100 mg | ¥ 1,643 | 现货 | ||
| 500 mg | ¥ 3,671 | 现货 | ||
| 1 mL * 10 mM (in DMSO) | ¥ 413 | 现货 | ||
| 产品描述 | Vandetanib (ZD6474) is a potent inhibitor of VEGFR2 (IC50: 40 nM). It also inhibits VEGFR3 and EGFR. |
| 靶点活性 | EGFR:500 nM (cell free), VEGFR3:110 nM (cell free), VEGFR2:40 nM (cell free) |
| 体外活性 | Vandetanib (ZD6474) is a potent inhibitor of KDR/VEGFR 2 tyrosine kinase activity (IC50: 40 nM). This compound has some additional activity versus the tyrosine kinase activity of VEGFR3 (IC50: 110 nM) and EGFR/HER1 (IC50: 500 nM). The activity of ZD6474 versus KDR tyrosine kinase translates into potent inhibition of VEGF-stimulated endothelial cell (human umbilical vein endothelial cell) proliferation in vitro (IC50: 60 nM) [1]. ZD6474 causes a dose-dependent inhibition of EGFR phosphorylation in mouse NIH-EGFR fibroblasts and human MCF-10A ras breast cancer cells. ZD6474 treatment resulted in a dose-dependent inhibition of soft agar growth in seven human cell lines with functional EGFR but lacking VEGFR-2. A dose-dependent supra-additive effect in growth inhibition and in apoptosis in vitro was observed by the combined treatment with ZD6474 and paclitaxel or docetaxel [2]. Vandetanib and neratinib displayed an inhibitory effect on the basal ABCG2-ATPase. At relatively high concentrations (10–20 mM), vandetanib inhibited the stimulated ABCG2-ATPase [3]. |
| 体内活性 | Administration of ZD6474 (2.5 mg/kg, i.v.) reversed a hypotensive change induced by VEGF (by 63%) but did not significantly affect that induced by basic fibroblast growth factor. Administration of 50 mg/kg/day ZD6474 (once-daily, p.o.) to athymic mice with intradermally implanted A549 tumor cells also inhibited tumor-induced neovascularization significantly (63% inhibition after 5 days). Histological analysis of Calu-6 tumors treated with 50 mg/kg/day ZD6474 for 24 days showed a significant reduction (>70%) in CD31 (endothelial cell) staining in nonnecrotic regions [1]. ZD6474 treatment of nude mice bearing palpable GEO colon cancer xenografts induced dose-dependent tumor growth inhibition. The antitumor activity of ZD6474 in GEO tumor xenografts was also found to be enhanced when combined with paclitaxel. Tumor regression was observed in all mice after treatment with ZD6474 plus paclitaxel, and it was accompanied by a significant potentiation in the inhibition of angiogenesis [2]. Vandetanib (15 mg/kg) had similar effects on the growth of H1650/PTEN and H1650 parental xenograft tumors [4]. |
| 激酶实验 | The ability of ZD6474 to inhibit the kinase activity associated with the VEGFRs KDR, Flt-1, and Flt-4 was determined using a previously described ELISA. Briefly, ZD6474 was incubated with enzyme, 10 mm MnCl2, and 2 μm ATP in 96-well plates coated with a poly(Glu, Ala, Tyr) 6:3:1 random copolymer substrate. Phosphorylated tyrosine was then detected by sequential incubation with a mouse IgG anti-phosphotyrosine 4G10 antibody, a horseradish peroxidase-linked sheep anti-mouse immunoglobulin antibody, and 2,2′-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid). Microcal Origin software was used to interpolate IC50 values by nonlinear regression. This methodology was adapted to examine selectivity versus tyrosine kinases associated with EGFR, PDGFRβ, Tie-2, FGFR1, c-kit, erbB2, IGF-IR, and FAK. All enzyme assays (tyrosine or serine-threonine) used appropriate ATP concentrations at or just below the respective Km (0.2–14 μm). Selectivity versus serine-threonine kinases (CDK2, AKT, and PDK1) was examined using a relevant scintillation proximity assay (SPA) in 96-well plates. CDK2 assays contained 10 mm MnCl2, 4.5 μm ATP, 0.15 μCi of [γ-33P]ATP/reaction, 50 mm HEPES (pH 7.5), 1 mm DTT, 0.1 mm sodium orthovanadate, 0.1 mm sodium fluoride, 10 mm sodium glycerophosphate, 1 mg/ml BSA fraction V, and a retinoblastoma substrate (part of the retinoblastoma gene, 792–928, expressed in a glutathione S-transferase expression system; 0.22 μm final concentration). Reactions were allowed to proceed at room temperature for 60 min before quenching for 2 h with 150 μl of a solution containing EDTA (62 mm final concentration), 3 μg of a rabbit immunoglobulin anti-glutathione S-transferase antibody and protein A SPA-polyvinyltoluene beads (0.8 mg/reaction). Plates were then sealed, centrifuged (1200 × g for 5 min), and counted on a Topcount NXT Microplate scintillation counter for 30 s [1]. |
| 细胞实验 | HUVEC proliferation in the presence and absence of growth factors was evaluated using [3H]thymidine incorporation. Briefly, HUVECs isolated from umbilical cords were plated (at passage 2–8) in 96-well plates (1000 cells/well) and dosed with ZD6474 ± VEGF or EGF (3 ng/ml) or bFGF (0.3 ng/ml). The cultures were incubated for 4 days (37°C; 7.5% CO2) and then pulsed with 1 μCi/well [3H]thymidine and reincubated for 4 h. Cells were harvested and assayed for the incorporation of tritium using a beta counter. IC50 data were interpolated as described above [1]. |
| 动物实验 | Methodology to enable blood pressure measurement in anesthetized rats was as described previously. Briefly, anesthesia was induced in male Alderley Park rats using α-chloralose by the i.v. route and then maintained with thiopentone via the i.p. route. Once surgical anesthesia was established, the carotid artery was cannulated to enable blood pressure recording using a pressure transducer and a Lectromed MT8P amplifier. The jugular vein was cannulated to allow growth factor administration. Body temperature was maintained with a thermostatically controlled heated blanket coupled to a rectal thermometer. Human VEGF165 (32 μg/kg) or bFGF (40 μg/kg) was administered as a bolus injection [0.1 ml/250 g body weight in 0.85% (w/v) sodium chloride], and a maximal blood pressure drop was recorded within 2 min (typically 26–30 mm Hg). These changes were sustainable for more than 20 min in control experiments. ZD6474 (2.5 mg/kg) or vehicle alone [25% (w/v) hydroxypropyl-β-cyclodextrin in Sorensons phosphate buffer (pH 5.5)] was administered i.v., and blood pressure was recorded 5 min later to determine the effect on growth factor-induced hypotension [1]. |
| 别名 | ZD6474, 凡德他尼 |
| 分子量 | 475.35 |
| 分子式 | C22H24BrFN4O2 |
| CAS No. | 443913-73-3 |
Powder: -20°C for 3 years | In solvent: -80°C for 1 year
DMSO: 27.5 mg/mL (57.85 mM), Sonication is recommended.
| 可选溶剂 | 浓度 体积 质量 | 1 mg | 5 mg | 10 mg | 25 mg |
| DMSO | 1 mM | 2.1037 mL | 10.5186 mL | 21.0371 mL | 52.5928 mL |
| 5 mM | 0.4207 mL | 2.1037 mL | 4.2074 mL | 10.5186 mL | |
| 10 mM | 0.2104 mL | 1.0519 mL | 2.1037 mL | 5.2593 mL | |
| 20 mM | 0.1052 mL | 0.5259 mL | 1.0519 mL | 2.6296 mL | |
| 50 mM | 0.0421 mL | 0.2104 mL | 0.4207 mL | 1.0519 mL |
对于不同动物的给药剂量换算,您也可以参考 更多...
请在以下方框中输入您的动物实验信息后点击计算,可以得到母液配置方法和体内配方的制备方法: 比如您的给药剂量是10 mg/kg,每只动物体重20 g,给药体积100 μL,一共给药动物10 只,您使用的配方为5% DMSO+30% PEG300+5% Tween 80+60% ddH2O。那么您的工作液浓度为2 mg/mL。
母液配置方法:2 mg 药物溶于 50 μL DMSO (母液浓度为 40 mg/mL), 如您需要配置的浓度超过该产品的溶解度,请先与我们联系。
体内配方的制备方法:取 50 μL DMSO 主液,加入 300 μL PEG300, 混匀澄清,再加 50 μL Tween 80,混匀澄清,再加 600 μL ddH2O, 混匀澄清。
您可能有的问题的答案可以在抑制剂处理说明中找到,包括如何准备库存溶液,如何存储产品,以及基于细胞的分析和动物实验需要特别注意的问题。
Vandetanib 443913-73-3 Angiogenesis Apoptosis Autophagy JAK/STAT signaling Tyrosine Kinase/Adaptors EGFR VEGFR inhibit ZD6474 Vascular endothelial growth factor receptor 凡德他尼 Inhibitor ZD-6474 ZD 6474 inhibitor
