store at low temperature | Powder: -20°C for 3 years | In solvent: -80°C for 1 year
Navitoclax (ABT-263) 是一种 Bcl-2 抑制剂,可与 Bcl-xL、Bcl-2、Bcl-w 等蛋白结合 (Ki<1 nM),具有有效性和口服活性。Navitoclax 具有抗肿瘤活性,可以诱导细胞凋亡。
| 规格 | 价格/CNY | 货期 | 数量 | |
|---|---|---|---|---|
| 1 mg | ¥ 233 | 现货 | ||
| 5 mg | ¥ 535 | 现货 | ||
| 10 mg | ¥ 797 | 现货 | ||
| 25 mg | ¥ 1,370 | 现货 | ||
| 50 mg | ¥ 1,980 | 现货 | ||
| 100 mg | ¥ 2,890 | 现货 | ||
| 200 mg | ¥ 3,960 | 现货 | ||
| 500 mg | ¥ 5,820 | 现货 | ||
| 1 g | ¥ 7,860 | 现货 | ||
| 1 mL * 10 mM (in DMSO) | ¥ 659 | 现货 | ||
| 产品描述 | Navitoclax (ABT-263) is a Bcl-2 inhibitor that binds to Bcl-xL, Bcl-2, and Bcl-w proteins (Ki<1 nM) with potent and oral activity. Navitoclax has antitumor activity and induces apoptosis. |
| 靶点活性 | BCL-W:<1 nM (Ki, cell free), BCL-XL:0.4 nM (Ki, cell free), BCL2:<1 nM (Ki, cell free) |
| 体外活性 |
方法:鼠原 B 淋巴细胞 FL5.12/Bcl-xL 和 FL5.12/Bcl-2 用 Navitoclax (0.001-1000 nmol/L) 处理 48 h,使用 CellTiter Glo 方法检测细胞活力。 结果:Navitoclax 逆转了 Bcl-2 或 Bcl-xL 的过表达所提供的保护 (EC50 分别为 60 和 20 nmol/L)。在 IL-3 存在的情况下,在 FL5.12 细胞不受促凋亡刺激的情况下,Navitoclax 诱导细胞死亡是无效的。[1] 方法:HCC 细胞 PLC/PRF/5、Hep3B、HepG2 和 Huh7 用 Navitoclax (5 μM) 处理 18 h,使用 Western Blot 方法检测靶点蛋白表达水平。 结果:在用 Navitoclax 治疗后,所有 HCC 细胞系中的 Mcl-1 水平显著增加,但 Bcl-2 和B cl-xL 水平没有显著变化。[2] |
| 体内活性 |
方法:为检测体内抗肿瘤活性,将 Navitoclax (100 mg/kg in 10% ethanol+30% polyethylene glycol 400+60% Phosal 50 PG) 口服给药给携带人 SCLC 和 ALL 异种移植物的 scid 小鼠,每天一次,持续二十一天。 结果:口服 Navitoclax 导致体内 SCLC 和 ALL 异种移植物肿瘤消退。[1] 方法:为检测体内抗肿瘤活性,将 Navitoclax (50-100 mg/kg in 10% ethanol+30% polyethylene glycol 400+60% Phosal 50 PG) 单剂量口服给药给携带人 SCLC 肿瘤 H146 的 scid 小鼠。 结果:单剂量 Navitoclax 治疗的 H146 肿瘤显示出大量的死亡和垂死细胞,包括血管化良好的肿瘤周围。[3] |
| 激酶实验 | ABT-737 and ABT-263 were synthesized as previously described. The enantiomer and BH3-only peptides were synthesized at Abbott. Binding affinities (Ki or IC50) were determined with competitive fluorescence polarization assays. The following peptide probe/protein pairs were used: f-bad (1 nmol/L) and Bcl-xL (6 nmol/L), f-Bax (1 nmol/L) and Bcl-2 (10 nmol/L), f-Bax (1 nmol/L) and Bcl-w (40 nmol/L), f-Noxa (2 nmol/L) and Mcl-1 (40 nmol/L), and f-Bax (1 nmol/L) and Bcl-2-A1 (15 nmol/L). Binding affinities for Bcl-xL were also determined using a time-resolved fluorescence resonance energy transfer assay. Bcl-xL (1 nmol/L, His tagged) was mixed with 200 nmol/L f-Bak, 1 nmol/L Tb-labeled anti-His antibody, and compound at room temperature for 30 min. Fluorescence was measured on an Envision plate reader using a 340/35 nm excitation filter and 520/525 (f-Bak) and 495/510 nm (Tb-labeled anti-His antibody) emission filters. Dissociation constants (Ki) were determined using Wang's equation [1]. |
| 细胞实验 | Human tumor cell lines were maintained at 37°C containing 5% CO2. SCLC cell lines were cultured in RPMI 1640 with 10% fetal bovine serum (FBS), 1% sodium pyruvate, 25 mmol/L HEPES, 4.5 g/L glucose, and 1% penicillin/streptomycin. Leukemia and lymphoma cell lines were cultured in RPMI 1640 supplemented with 10% FBS and 1% penicillin/streptomycin. Cells (1 × 10^4–5 × 10^4) were treated for 48 h in 96-well culture plates in a final volume of 100 μL and cytotoxicity was assessed with the CellTiter Glo assay [1]. |
| 动物实验 | C.B.-17 scid-bg or C.B.-17 scid mice were implanted with 5 × 10^6 (1 × 10^6 for DoHH2) cells in 0.2 mL 50% Matrigel s.c. into the right flank. Tumor-bearing mice were size matched (~235 mm3; day 0) into treatment and control groups, ear tagged, and monitored individually. Tumor volume was measured two to three times weekly by electronic calipers (volume = length × width2 / 2). Tumor growth inhibition was calculated based on the difference in mean tumor volumes between treated and appropriate vehicle control groups. Partial response (PR) is defined as ≥50% tumor growth inhibition, and complete response (CR) is defined as nonpalpable tumor. All studies used 8 to 10 mice per group. ABT-263 was formulated in 10% ethanol, 30% polyethylene glycol 400, and 60% Phosal 50 PG and administered p.o. The other agents used [rituximab, doxorubicin, cyclophosphamide, vincristine, bortezomib, and prednisone] were administered i.p., p.o., or i.v. and formulated according to the manufacturers' recommendations. For combination studies, ABT-263 was given ~2 h before the other agents, except bortezomib, which was given ~4 h before ABT-263 [1]. |
| 别名 | ABT-263 |
| 化合物与蛋白结合的复合物 |
Bcl_2-Navitoclax (ABT-263) Complex |
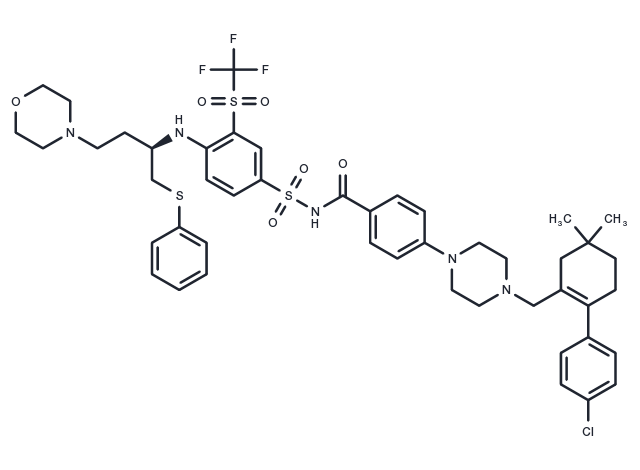
| 分子量 | 974.61 |
| 分子式 | C47H55ClF3N5O6S3 |
| CAS No. | 923564-51-6 |
store at low temperature | Powder: -20°C for 3 years | In solvent: -80°C for 1 year
Ethanol: < 1 mg/mL (insoluble or slightly soluble)
DMSO: 200 mg/mL (205.21 mM), Sonication is recommended.
H2O: < 1 mg/mL (insoluble or slightly soluble)
| 可选溶剂 | 浓度 体积 质量 | 1 mg | 5 mg | 10 mg | 25 mg |
| DMSO | 1 mM | 1.0261 mL | 5.1303 mL | 10.2605 mL | 25.6513 mL |
| 5 mM | 0.2052 mL | 1.0261 mL | 2.0521 mL | 5.1303 mL | |
| 10 mM | 0.1026 mL | 0.513 mL | 1.0261 mL | 2.5651 mL | |
| 20 mM | 0.0513 mL | 0.2565 mL | 0.513 mL | 1.2826 mL | |
| 50 mM | 0.0205 mL | 0.1026 mL | 0.2052 mL | 0.513 mL | |
| 100 mM | 0.0103 mL | 0.0513 mL | 0.1026 mL | 0.2565 mL |
对于不同动物的给药剂量换算,您也可以参考 更多...
请在以下方框中输入您的动物实验信息后点击计算,可以得到母液配置方法和体内配方的制备方法: 比如您的给药剂量是10 mg/kg,每只动物体重20 g,给药体积100 μL,一共给药动物10 只,您使用的配方为5% DMSO+30% PEG300+5% Tween 80+60% ddH2O。那么您的工作液浓度为2 mg/mL。
母液配置方法:2 mg 药物溶于 50 μL DMSO (母液浓度为 40 mg/mL), 如您需要配置的浓度超过该产品的溶解度,请先与我们联系。
体内配方的制备方法:取 50 μL DMSO 主液,加入 300 μL PEG300, 混匀澄清,再加 50 μL Tween 80,混匀澄清,再加 600 μL ddH2O, 混匀澄清。
您可能有的问题的答案可以在抑制剂处理说明中找到,包括如何准备库存溶液,如何存储产品,以及基于细胞的分析和动物实验需要特别注意的问题。
Navitoclax 923564-51-6 Apoptosis BCL inhibit Inhibitor Bcl-2 Family ABT-263 ABT 263 ABT263 inhibitor
